One Platform. Every Post-Market Workflow. AI Across Everything.
Smarteeva takes a fundamentally different approach. We built one unified platform where every product shares the same data model, the same AI engine, the same audit trail, and the same Salesforce.
Traditional Platforms
With Smarteeva
Six Integrated Products. One Connected Ecosystem.
Start with one product. Add more when you're ready. Every product runs on the same data model and AI engine, so a complaint record created in Complaints Management is already connected to your Adverse Events, PMS, and Recall workflows. No migration. No reintegration. No "Phase 2" project.
Smarteeva Orchestra
The full complaint lifecycle in one guided workflow - from the moment a complaint arrives to the day it closes.
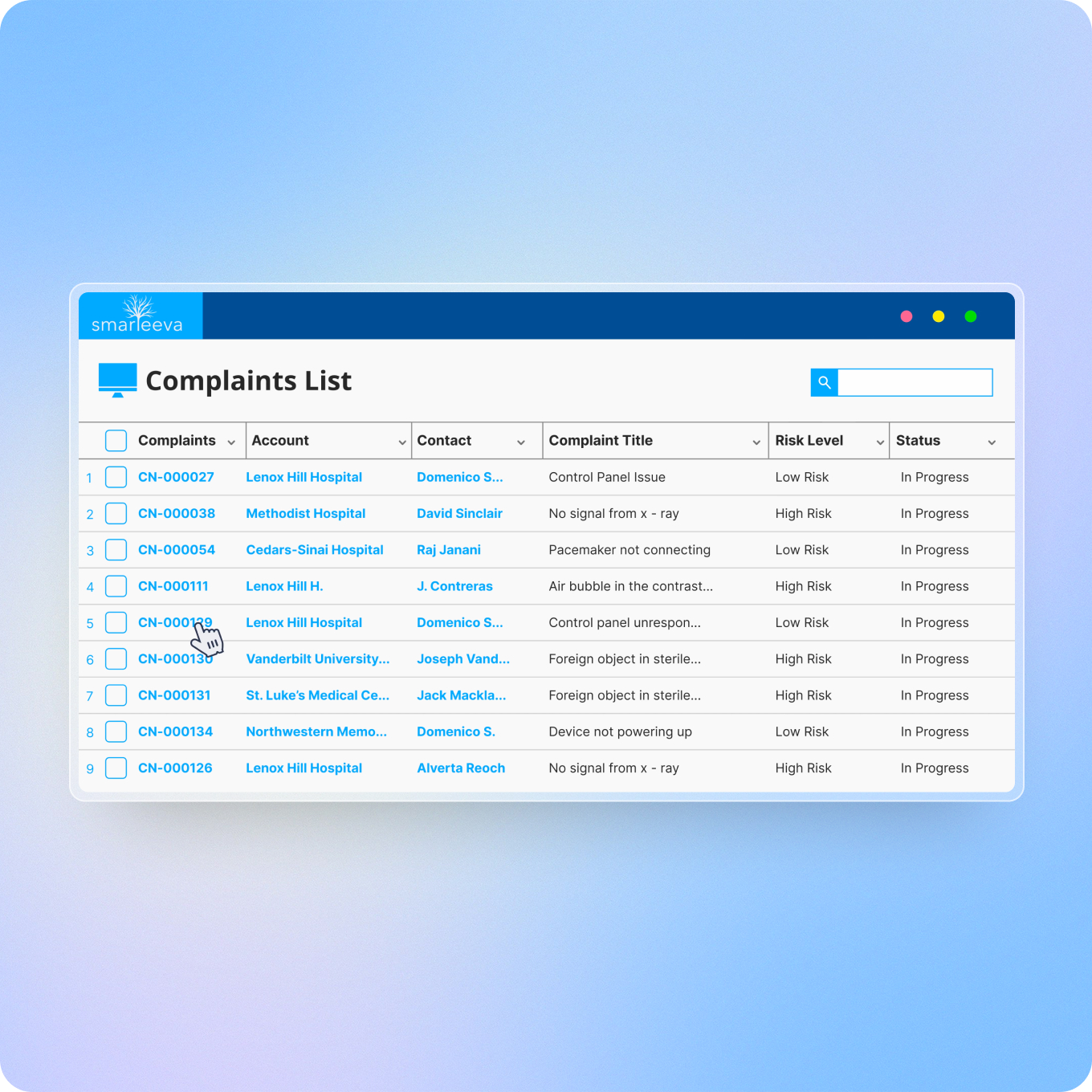
Email, manual entry, and bulk import all feed the same structured process. AI extracts fields from unstructured emails, flags potential duplicates using similarity logic (not just exact matches), and suggests IMDRF codes from the complaint narrative. Every record carries a full audit trail. Every investigation, MDR, and CAPA links back to the source complaint automatically.
Whether your team handles 50 complaints a month or 5,000, the workflow is the same. The AI is the same. The audit trail is the same.

Complaints Management
Next-generation complaint handling with AI-driven intake, automated risk assessment, machine learning-guided investigation, and customer self-service - compliant with FDA, EU MDR, PMDA, and TGA.
Adverse Events
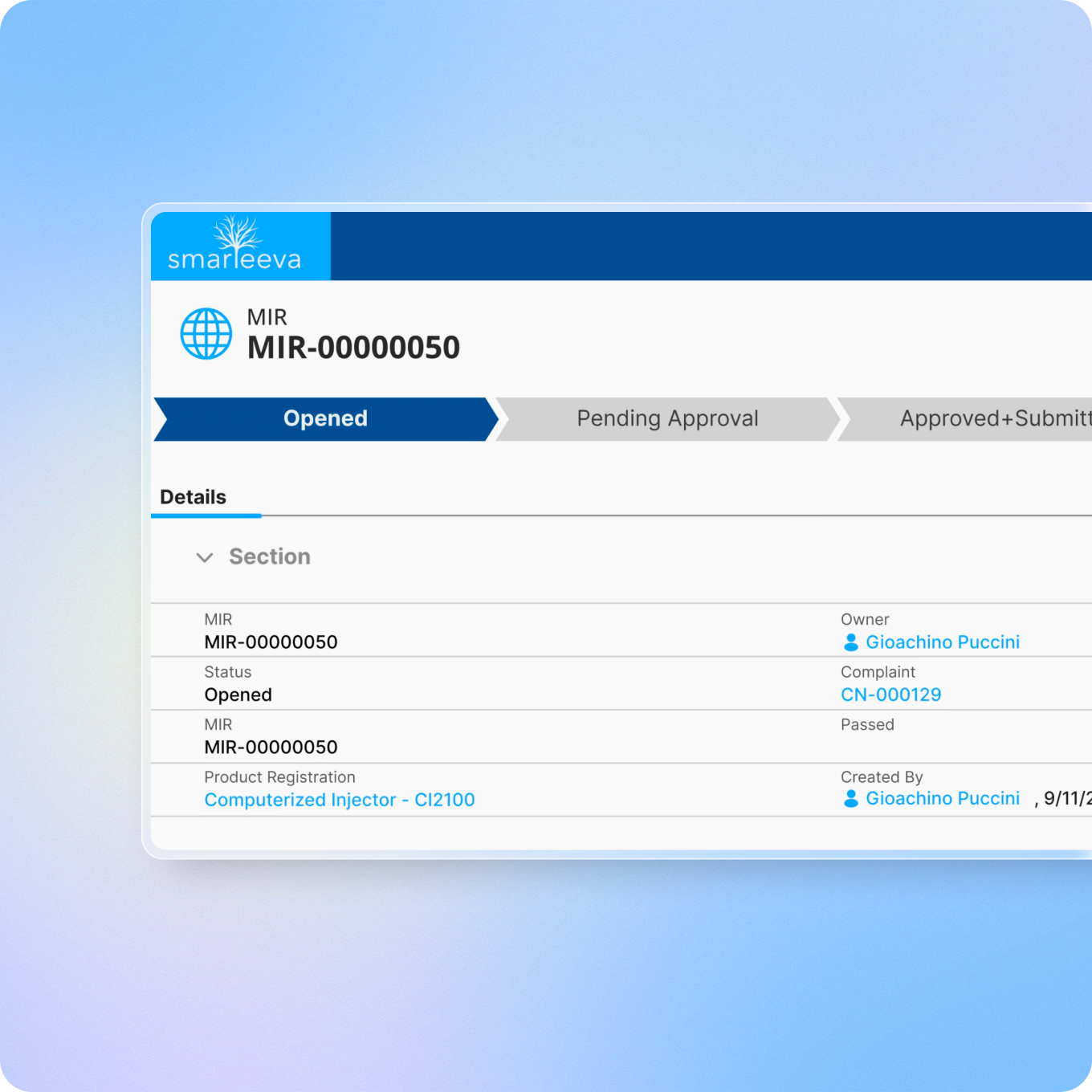
AI-powered adverse event detection from complaint narratives, automated MedWatch and MDR report generation, and seamless tracking of MDRs, MIRs, CVRs, and PSURs, with smart audit trails and e-signatures

Automated PMS Reporting
Fully automated post-market surveillance reports - EU PSUR, UK PSUR, EU PMSR, UK PMSR, PMPF-ER, and HCSR, generated in minutes by pulling directly from your complaint and quality data
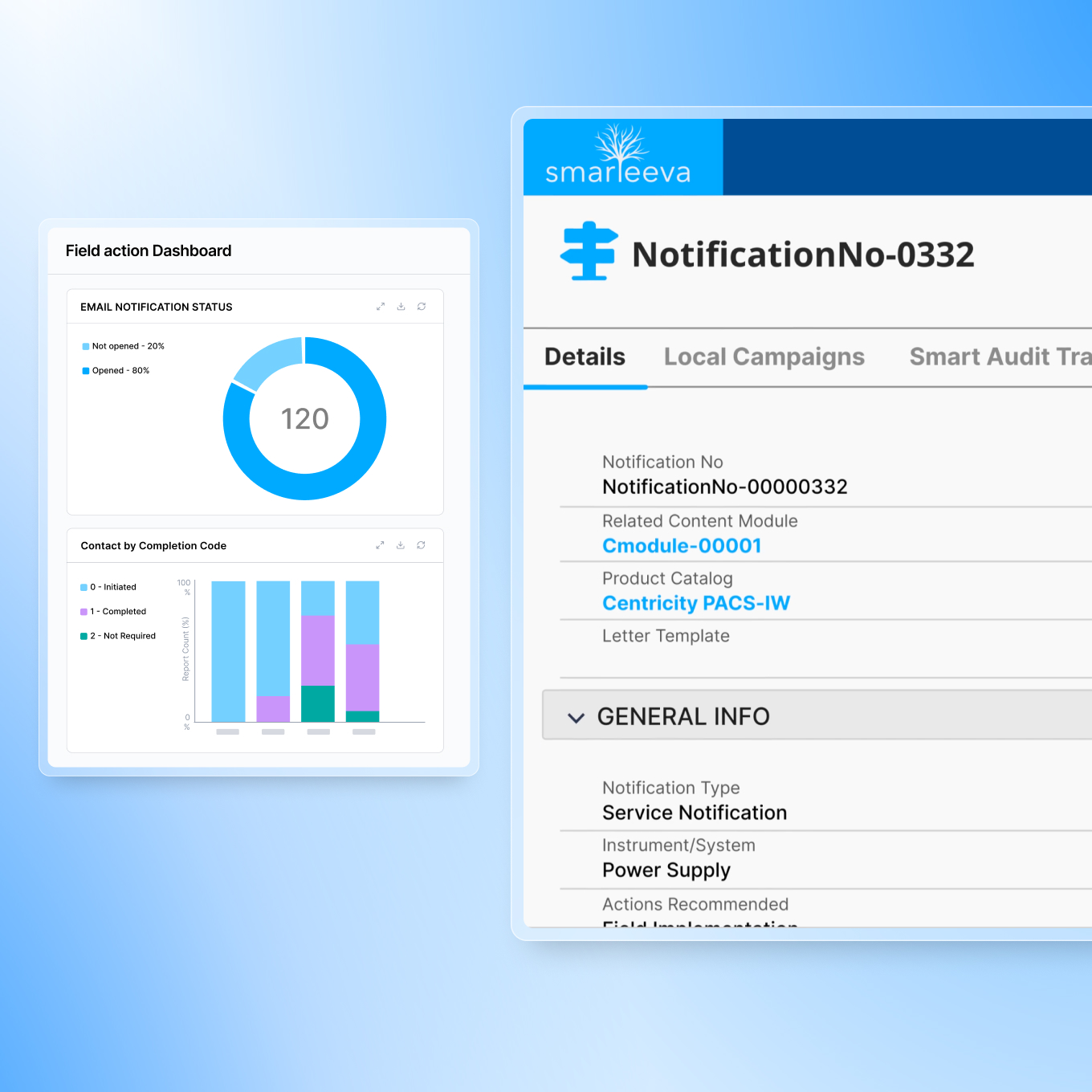
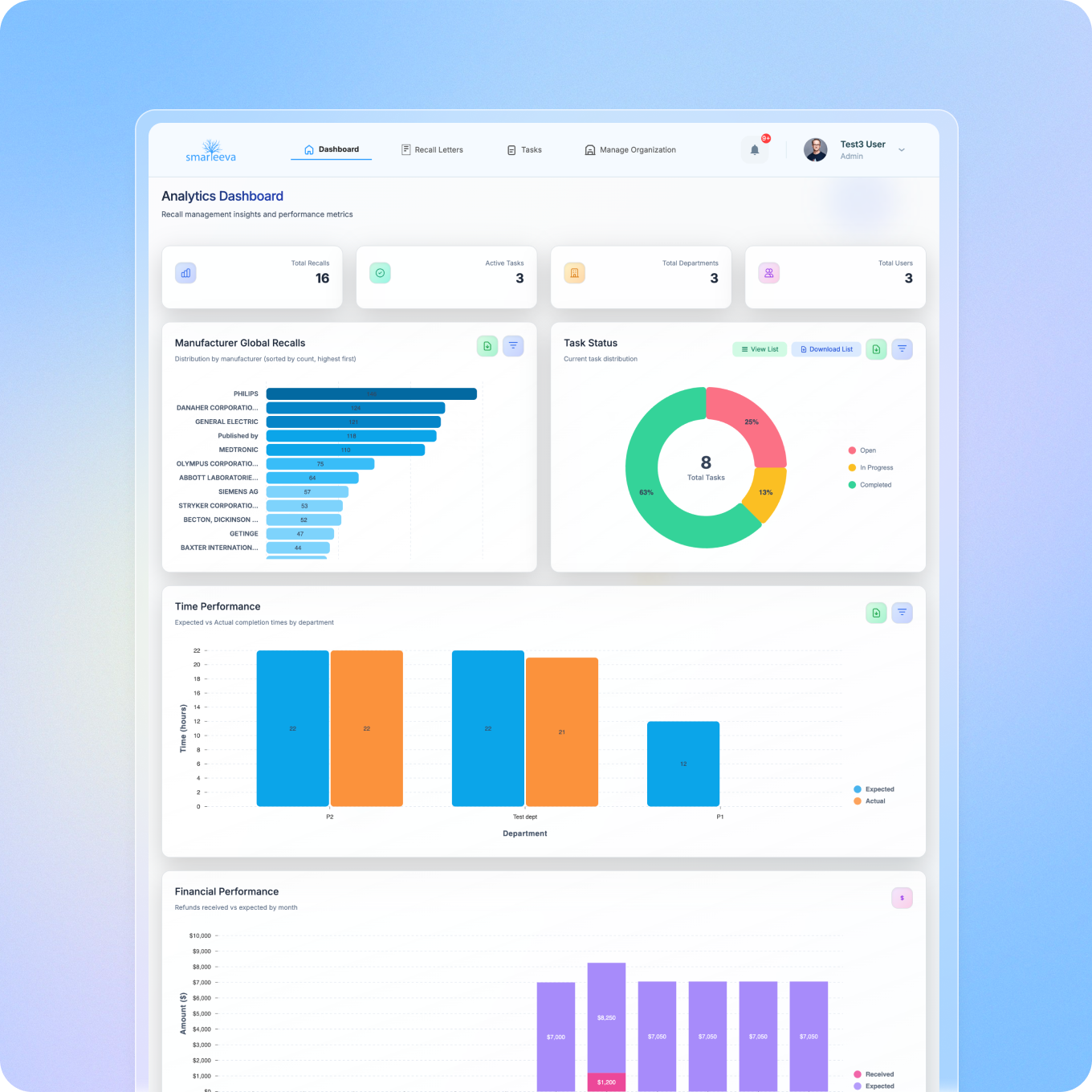
Recall Management
Fast notification generation, intelligent approval workflows, follow-up tracking, and a dedicated recall management portal for customers, affiliates, and distributors.
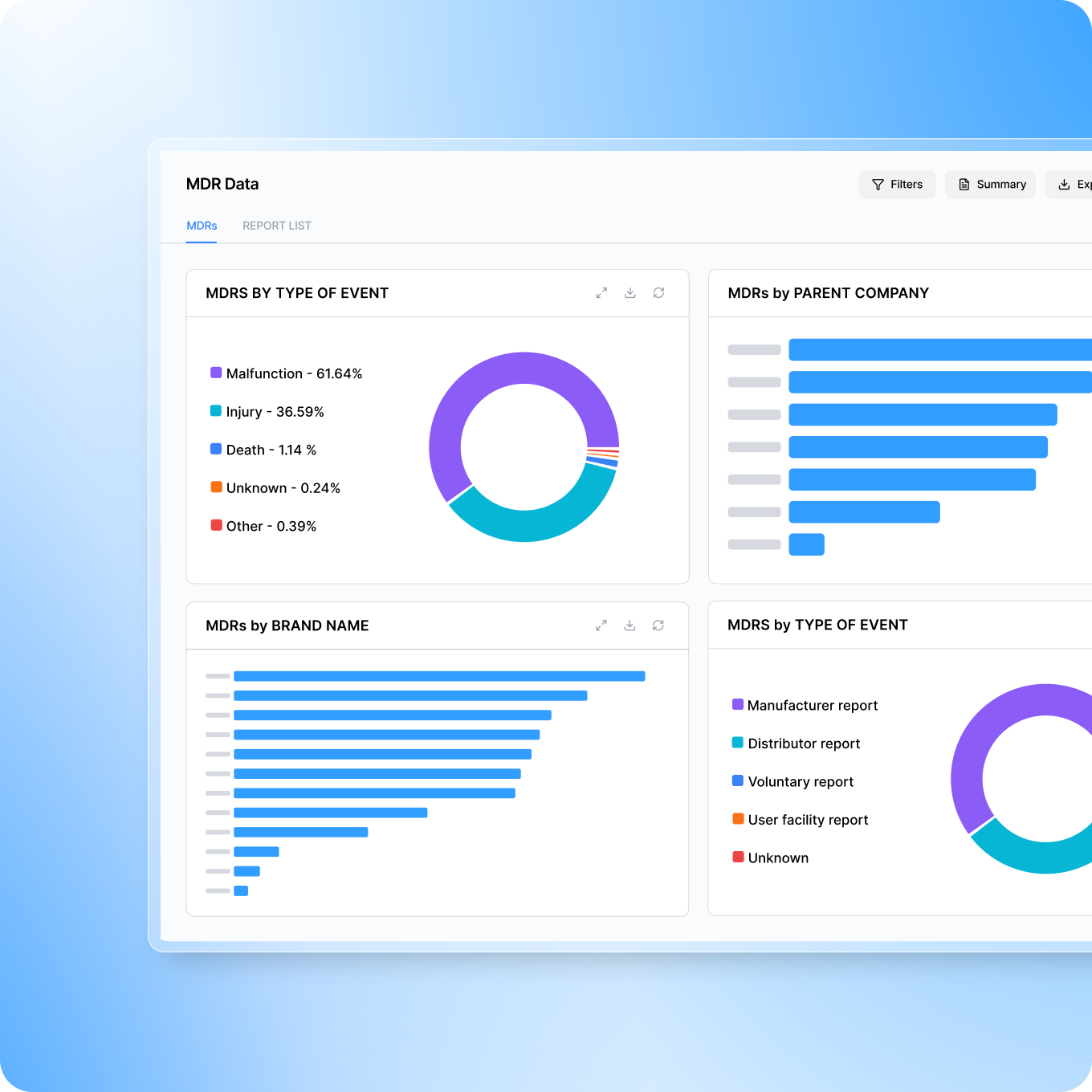
MDREngine
Aggregated, cleaned, and correlated regulatory data - MDRs, FAERs, recalls, warning letters, clinical studies, and industry news, in one searchable intelligence platform.
MedTech Recalls Gateway
A two-sided portal connecting medical device manufacturers with the hospitals and health systems affected by their recalls. Manufacturers issue recall notifications through one dashboard. Health systems receive alerts, acknowledge receipt with one click, and track required actions, no email chains, no lost PDFs, no manual follow-up spreadsheets.
AI That Runs Through Everything
Smarteeva's AI continuously monitors complaint trends and adverse event patterns, flagging emerging safety signals before they become regulatory incidents or recalls.
Salesforce & Secure Cloud Infrastructure
Smarteeva runs natively on Salesforce and is powered by secure, globally distributed cloud infrastructure. Your data stays in the same environment as your CRM and Service Cloud, your team works in a familiar interface, and IT manages a single platform instead of multiple systems.
Enterprise-grade compute, storage, and global availability ensure your post-market surveillance data is always accessible, always fast, and always secure, wherever your teams operate. Together, Salesforce and cloud-native architecture give Smarteeva the security, compliance, and scalability that standalone quality management systems can't match.
Salesforce-Native
Cloud-Powered
Regulatory-Grade Security
Configurable, Not Coded
Configurable, Not Coded
Configurable, Not Coded
Built for the Teams That Own Quality






