Smarteeva Orchestra:
Your Team Builds the AI. No Code. No Data Scientists.
Orchestra is a visual drag-and-drop canvas where Quality and Regulatory teams design AI agent workflows tailored to their specific devices, regulations, and processes.

Orchestra powers the AI behind every capability on this page. But it's also a builder. If your team has a workflow that's unique to your organization. A specific triage logic, a custom escalation path, a validation check nobody else needs, they can build an agent for it. In minutes. Without writing code.
Choose your LLM provider
& connect your data sources.

Define guardrails and
validation rules.
Deploy agents that run inside your
existing Salesforce workflow
AI at Every Stage.
From Intake to Submission
Smarteeva's AI is embedded across six critical stages of the post-market surveillance lifecycle. At each stage, AI handles the data-intensive work that slows humans down, while your team retains full control over every quality and regulatory decision.
Complaint Intake & Classification
Complaints arrive from email, phone, web forms, and bulk imports, each in a different format, each missing different fields. Smarteeva AI reads unstructured complaint text, extracts product, patient, event, and reporter details, and creates a structured record automatically. Missing fields trigger a targeted follow-up, not a generic email chain.
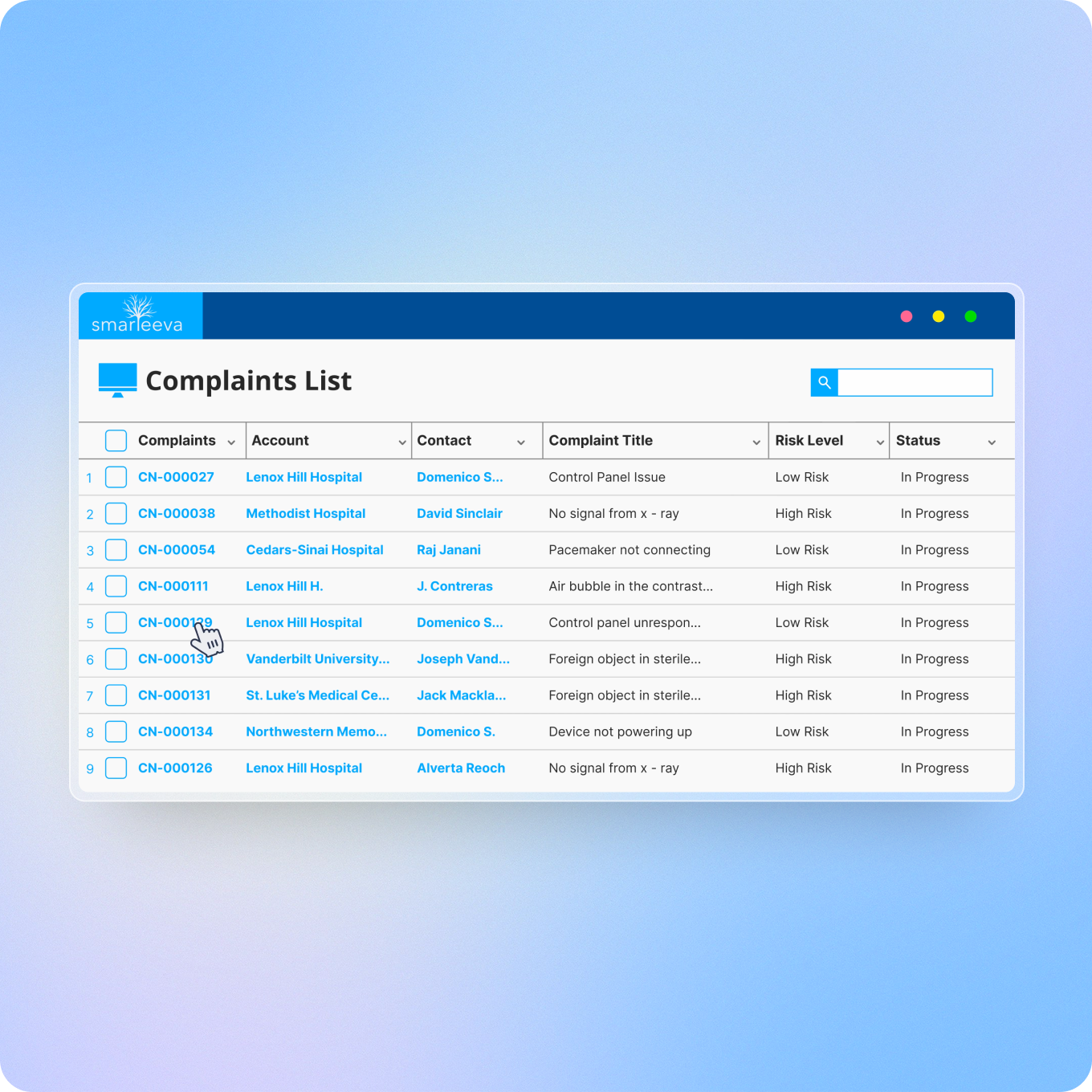
AI-Powered Triage & Prioritization
Complaint queues often mix critical and routine issues, forcing manual triage. Smarteeva AI instantly classifies and prioritizes complaints so high-risk cases are escalated immediately.
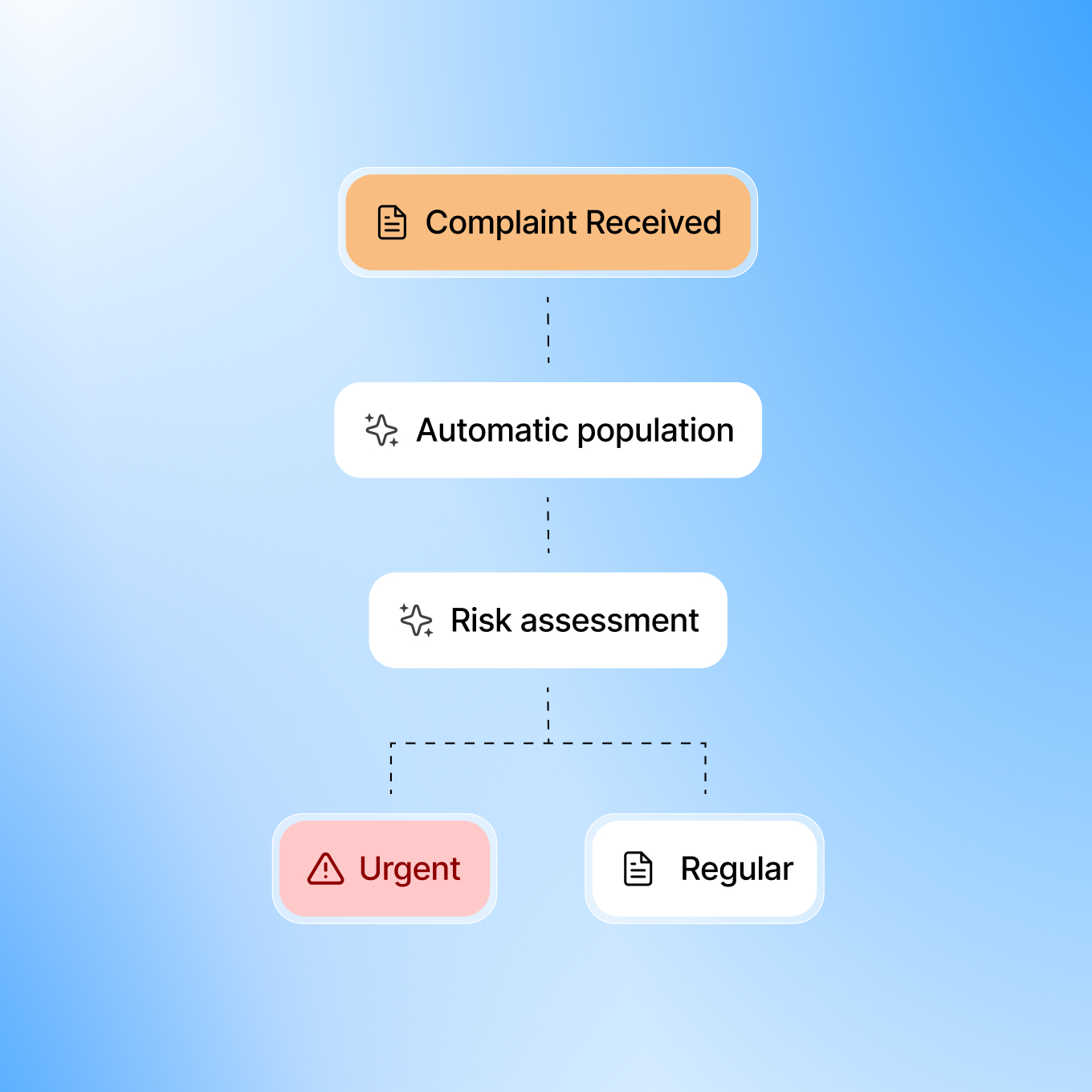
Adverse Event Detection from Complaint Narratives
Adverse events can be hidden in vague complaint narratives and missed during manual review. Smarteeva AI scans every complaint to detect potential adverse events and flags them instantly for regulatory evaluation.

AI-Assisted Investigation
Investigations often start with blank forms and manual data gathering. Smarteeva AI surfaces similar past cases, suggests investigation paths, and pre-populates records to speed up root-cause analysis.

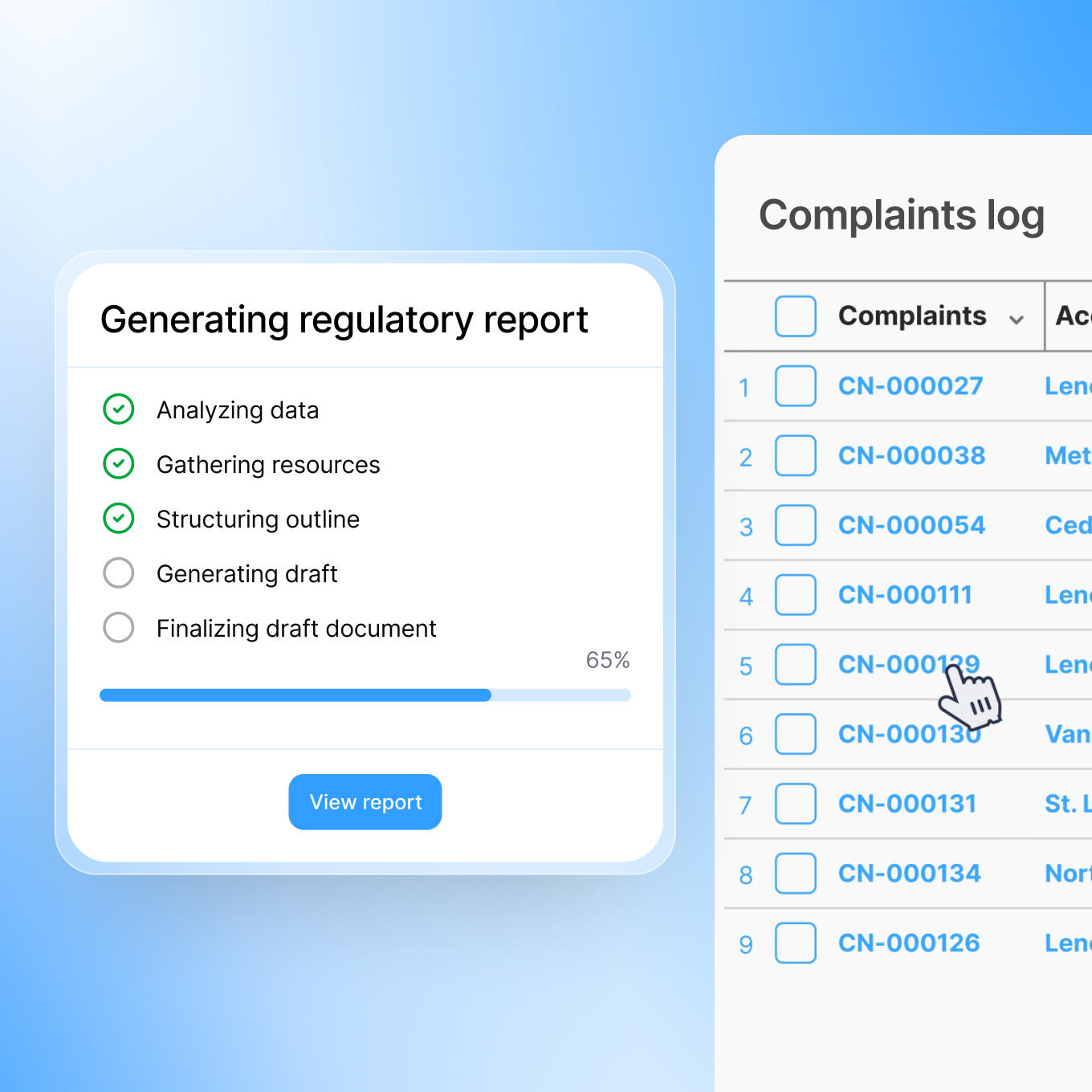
Automated Regulatory Reporting
Regulatory reports typically require extensive manual compilation. Smarteeva automatically generates draft submissions and surveillance reports from your data, ready for regulatory review.
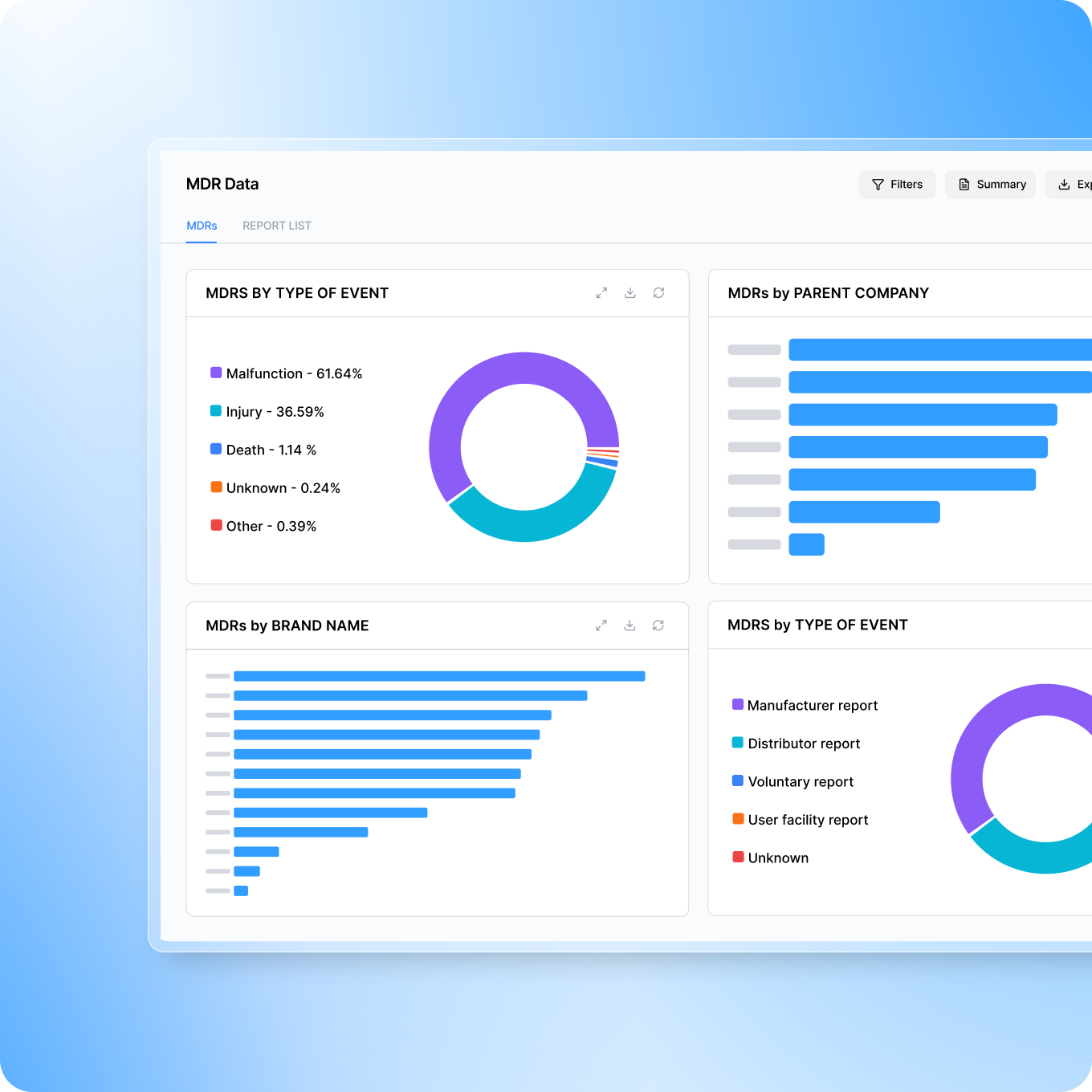
Predictive analytics & Signal detection
Quality trends are often discovered too late. Smarteeva AI analyzes complaint data in real time to detect emerging patterns, anomalies, and potential safety signals early.