MDREngine
FDA Data at Your Fingertips - MDRs, Warning Letters, Recalls, and FAERs
MDREngine aggregates, cleans, correlates, and enhances medical device regulatory data from the FDA and other sources, giving your quality, regulatory, and R&D teams instant access to the intelligence they need.
- Search MDRs, recalls, and warning letters instantly
- Cross-referenced data, not fragmented databases
- Always current, never a stale snapshot
FDA databases are sprawling, inconsistently formatted, and difficult to search. Data from different sources doesn't cross-reference. By the time you've compiled what you need, hours or days have passed.
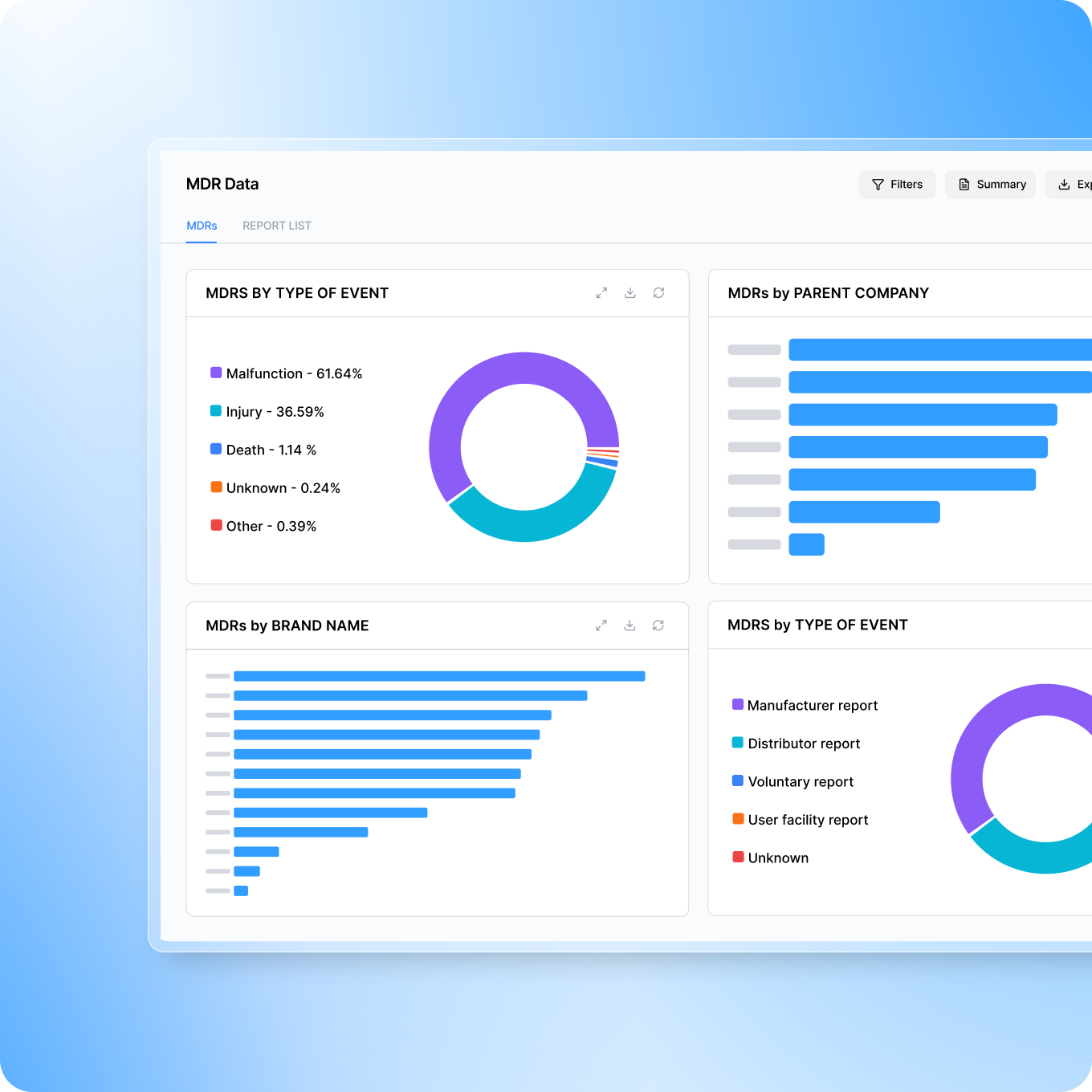
One platform. Every data source. Fully correlated.
Competitive intelligence
How do your complaint rates compare to industry benchmarks? MDREngine pulls adverse event history across manufacturers and device categories so you always know where you stand.
Submission support
What adverse event history exists for similar devices? Pull the data you need for regulatory submissions without manually searching MAUDE.
Product intelligence
What failure modes are being reported in your device category? MDREngine surfaces signals that inform next-generation design decisions.
What's inside the data lake
Medical Device Reports (MDRs)
The complete database of adverse event reports filed with the FDA, cleaned and enhanced for searchability. Search by device type, manufacturer, event type, patient outcome, and more.
FDA Adverse Event Reports (FAERs)
Adverse event data for drugs and combination products, cross-referenced with device data for companies operating across product categories.
Recalls
FDA recall data including Class I, II, and III recalls, with details on affected products, recall scope, corrective actions, and manufacturer responses.
Warning Letters
FDA warning letters indexed by company, product category, and violation type. Understand enforcement trends and identify common compliance gaps in your product category.
Clinical Studies
Clinical study data relevant to medical devices, providing context for regulatory submissions and post-market surveillance reports.
Industry News
Curated news and regulatory updates affecting the medical device industry, keeping your team informed of developments that could impact your products.
Use cases
Competitive Benchmarking
How do your adverse event rates compare to similar devices from other manufacturers? MDREngine lets you benchmark your quality data against industry-wide FDA data.
PMS Report Support
PMS reports under EU MDR require companies to consider external data sources. MDREngine provides the literature and regulatory data that supports your PMS report conclusions.
Regulatory Submission Preparation
When preparing 510(k) submissions, PMA supplements, or other filings, MDREngine provides instant access to adverse event history, recall history, and clinical data for predicate and comparable devices.
Signal Detection
Identify emerging safety signals across the industry that could affect your products, before they show up in your own complaint data.
Audit Preparation
When an FDA inspector asks about adverse event trends in your product category, MDREngine gives you the data to contextualize your own quality performance within the broader industry landscape.








